|
Phospho-Protein Phosphatase Inhibitor-1
(Catalog # P025 & P026)
Description:
Human recombinant protein phosphatase inhibitor-1 is produced from E. coli.,
purified from freshly extracted E. coli lysates, and purified to
homogeneity using methods developed by LAE Biotechnology Co., LTD. The final fraction of protein phosphatase
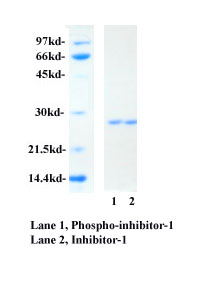
inhibitor-1 exhibits a single polypeptide band of 27 kDa. Protein phosphatase inhibitor-1 can be
phosphorylated by cAMP-dependent protein kinase. Protein phosphatase inhibitor-1 after phosorylation
on Thr-35 becomes a potent inhibitor of type 1 protein phosphatase (1, 2). Phospho-protein phosphatase inhibitor-1 specifically inhibits the catalytic subunit of type 1
protein phosphatases (PP1) at nanomolar concentrations (IC50 ~ 2 nM). Protein phosphatase
inhibitor-1 was prepared by phosphorylation on Thr-35 by cAMP-dependent protein
kinase.
Quality of Phospho-Protein Phosphatase Inhibitor-1:
1X PP1 Reaction Buffer:
50 mM Tris-HCl, 5 mM dithiothreitol,
0.1 mM EDTA, pH 7.0
Reaction Conditions:
1X PP1 Reaction Buffer supplemented with
1 mM MnCl2 and 5 mM Caffeine
Reaction should be performed at 30 C.
Supply:
Protein phosphatase inhibitor-1 is supplied in lyophilized form at the dose of 100 ug per vail (Catalog # P025) and
at the dose of 50 ug per vail (Catalog # P026).
Reconstitution:
Lyophilized protein phosphatase inhibitor-1 (catalog # P025)
should be reconstituted in 100 ul of 50 mM Tris-HCl, pH 7.0, while lyophilized
protein phosphatase inhibitor-1 (catalog # P026) should be reconstituted in 50
ul of 50 mM Tris-HCl, pH 7.0, to make a final concentration of 1 mg/ml.
Shipping/Storage:
The protein phosphatase inhibitor-1 is shipped on ice and must
be stored at -20 C or lower.
Legal consideration: FOR RESEARCH USE ONLY
Notes:
1. Supplement with 1 mM MnCl2 and 5 mM caffeine
(required only if phosphorylase a is the substrate).
2. Protein phosphatase inhibitor-1 has been purified
to > 95% homogeneity as determined by SDS-PAGE and Coomassie Blue staining.
3. Recommended long term storage -70 C; avoid repeated freeze/thaw cycles.
Quality Assurance Statement:
Protein phosphatase inhibitor-1 contains no
detectable protease activity. Tests for phosphatase activity showed no
detectable activity.
References:
1. Connor, J. H. (1998) Inhibitor-1 interaction domain that mediates the inhibition of protein phosphatase-1. J. Biol. Chem.
273, 27716.
2. Connor, J. H. (1999) Importance of the
beta12-beta13 loop in protein phosphatase-1 catalytic subunit for inhibition by
toxins and mammalian protein inhibitors. J. Biol. Chem. 274, 22366.
| 




